RDPA 2025: Strategies for Robust HPLC Method Transfer: The Critical Role of Column Chemistry and Dwell Volume
RDPA 2025
Recent Developments in Pharmaceutical Analysis
2-5 September 2025, Pavia, Italy
Poster Scientifico
Strategies for Robust HPLC Method Transfer:
The Critical Role of Column Chemistry and Dwell Volume
M.Maiocchi – Product Manager CPS Analitica
ABSTRACT
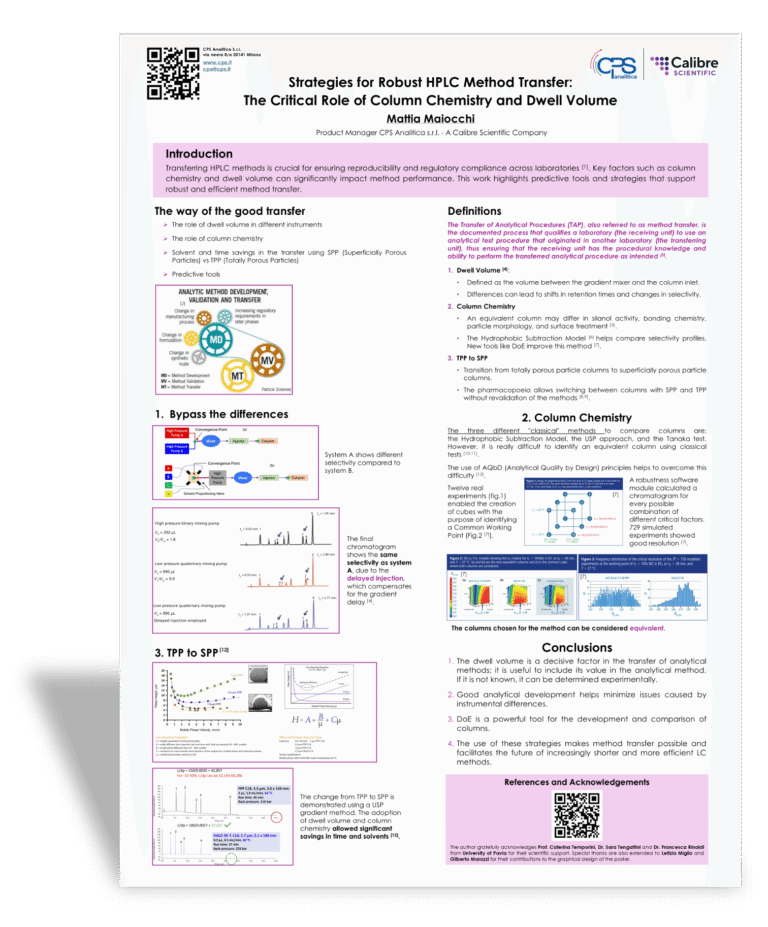
Transferring HPLC methods is a pivotal step in pharmaceutical and analytical workflows, with direct implications for reproducibility, accuracy, and regulatory compliance across different laboratories and instruments. Among the Critical Method Parameters (CMPs), column chemistry and dwell volume play a particularly decisive role.
Even among columns marketed as equivalent, variations in stationary phase characteristics—such as silanol activity, bonding chemistry, particle morphology, and surface treatment—can significantly affect retention behavior and selectivity. Tools like the Hydrophobic Subtraction Model (Snyder et al., 2011) provide a framework to assess column selectivity profiles, helping ensure compatibility and reducing risks during method transfer.
Dwell volume, defined as the volume between the gradient mixer and the column inlet, is equally critical in gradient elution. Differences between instruments can cause shifts in retention times and altered selectivity. Jaekel et al. (2023) highlighted that unadjusted dwell volume mismatches can severely impact performance, even under identical gradient programs. Earlier, Schellinger and Carr (2005) proposed practical adaptations, such as gradient start-time adjustments and modification of delay volumes, to facilitate smoother method transfer.
Moreover, in the poster there will be a space for the passage of TPP column to SPP in order to allow a tranfer with saving of solvent and time (Shuster 2023).
In summary, a systematic and predictive approach to method transfer—comprising column equivalency verification, dwell volume adjustment, and gradient re-optimization—enhances method robustness and aligns with regulatory standards (e.g., ICH Q2(R2), USP <1224>), promoting reproducible and compliant HPLC performance across platforms.
References:
- Snyder, L., Dolan, J., Marchand, D. e Carr, P., (2012). The Hydrophobic-Subtraction Model of Reversed-Phase Column Selectivity. In: Advances in Chromatography [online]. CRC Press. pp. 297–376. doi: https://doi.org/10.1201/b11636
- Jaekel, A., Wirtz, M., Lamotte, S., & Legelli, M. (2023). Performance in (Ultra‐)HPLC – How to qualify and optimize instruments in practice. Journal of Separation Science, 2200894. https://doi.org/10.1002/jssc.202200894
- Schellinger, A. P., & Carr, P. W. (2005). A practical approach to transferring linear gradient elution methods. Journal of Chromatography A, 1077(2), 110–119. https://doi.org/10.1016/j.chroma.2005.04.088
- Schuster, S., Time and Solvent Savings by Modernizing USP Methods with HALO® Columns Following the New <621> Guidelines, HALO® Technical White Paper, Advanced Materials Technology, 2023, WP_2023_621-USP_FINAL,pp.1–6. https://halocolumns.com/wp content/uploads/2023/06/WP_2023_621-USP_FINAL.pdf
REFERENCES
[1] Guillarme, D.; Veuthey, J.-L.; Schappler, J.; Rudaz, S., Method transfer for fast liquid chromatography in pharmaceutical analysis: Application to short columns packed with small particles. Part II: Gradient experiments, J. Chromatogr. A 2008, 1189, 3–217
[2] Particle Sciences, The Central Role Of Analytic Method Development And Validation In Pharmaceutical Development, Particle Sciences Technical Brief 2010, https://particlesciences.com/wp-content/uploads/2019/10/analytic_method_development_in_pharmaceutical.pdf
[3] United States Pharmacopeia, <1224> Transfer of Analytical Procedures, USP 48 – NF 43, United States Pharmacopeial Convention, Rockville, MD, 2025.
[4] D.R. Stoll, The gradient delay volume, Part I: Theory, LCGC Int. 2024, 37 (4), 6–10. https://doi.org/10.56530/lcgc.int.wj6080e9
[5] Bell, D. S.; Green, A. I., What Is on Your HPLC Particle? A Look at Stationary Phase Chemistry and Synthesis, LCGC Asia Pacific, 2021, 24(1), 28–33. https://www.chromatographyonline.com/view/hplc-particle-a-look-at-stationary-phase-chemistry-synthesis
[6] Snyder, L. R.; Dolan, J. W.; Carr, P. W., The hydrophobic-subtraction model of reversed-phase column selectivity, J. Chromatogr. A, 2004, 1060(1–2), 77–116. https://doi.org/10.1016/j.chroma.2004.08.121
[7]Molnár, I.; Rieger, H.-J., Equivalent Column Selection in HPLC, LCGC Europe, 2017, 30(11), 13–17. https://www.chromatographyonline.com/view/equivalent-column-selection-hplc-0
[8] European Pharmacopoeia, 2.2.46 Chromatographic Separation Techniques, Ph. Eur. 12.0 [online], Council of Europe – EDQM, Strasbourg, France, 2025. Available at: https://pheur.edqm.eu
[9] United States Pharmacopeia, <621> Chromatography, USP 48 – NF 43 [online], United States Pharmacopeial Convention, Rockville, MD, 2025. Available at: https://www.uspnf.com
[10] Guillarme Davy, Veuthey Jean‑Luc, Meyer Veronika R., Method Transfer in HPLC, LCGC Eur. 2008, 21 (6), 322–327
[11]Enesei, I. Kapui, S. Fekete, R. Kormány, Updating the European Pharmacopoeia impurity profiling method for terazosin and suggesting alternative columns, J. Pharm. Biomed. Anal. 2020, 187, 113371.
[12] Schuster, S., Time and Solvent Savings by Modernizing USP Methods with HALO® Columns Following the New <621> Guidelines, HALO® Technical White Paper, Advanced Materials Technology, 2023, WP_2023_621-USP_FINAL, pp. 1–6. https://halocolumns.com/wp-content/uploads/2023/06/WP_2023_621-USP_FINAL.pdf